SOIL CHEMISTRY
Soils transport and move water, provide homes for thousands of bacteria and other creatures, and have many different arrangements of weathered rock and minerals. When soils and minerals weather over time, the chemical composition of soil also changes. However, nothing changes the chemistry of soils faster than humans do.
Many of today’s soil chemistry problems have to do with environmental sciences. What happens when a chemical is accidentally spilt in the soil? How fast does it break down? What does it break down into? Where does it go and how fast does it move? A soil chemist may ask these example questions. Soil chemists research concerns about organic and inorganic soil contamination, pesticides and other pollutants, and environmental health risks.
How does soil keep fertilizers from leaching out of the plant root zone where they are needed?
Soils are composed of solid particles with spaces (pores) between them. The pores always contain some air and water. The solids can be minerals or organic (decomposed plant and animal remains). Soil particles are classified by size: sands (2.0 to 0.05 mm), silts (0.05 to 0.002 mm) and clays (<0.002 mm). Due to their small size, clays particles have thousands of times more surface area than sands – and clay surfaces have charges.
It may seem confusing, but in soil science, clay can refer to the particle size (<0.002 mm) or to a type of mineral. There are different kinds of clay minerals, which have varying amounts of negative surface charge. Organic matter also has a charge. These charges affect the ability of soil to hold and release plant nutrients. Plant nutrients are ions - atoms, compounds and molecules with a net electrical charge. Ions have negative charges and cations have positive charges. Water in soil always contains ions. These ions can freely move to and from the water and the charged soil surfaces in a process called ion exchange.
Both clay and organic matter typically have negative charges, though some might develop positive charges in strongly acidic soils.
Ion Exchanges
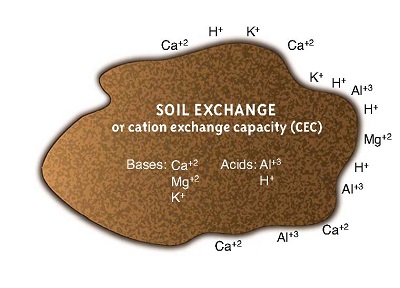
This diagram represents soil cations attached firmly to the soil.
Ion exchange involves the movement of cations (positively charged elements like calcium, magnesium, and sodium) and anions (negatively charged elements like chloride, and compounds like nitrate) through the soils. In the United States, cation exchange is much more common.
Cation exchange is the interchanging between a cation in the solution of water around the soil particle, and another cation that is stuck to the clay surface. The number of cations in the soil water solution is much smaller than the number that is attached to soil particles.
The total amount of positive charges that the soil can absorb is called the cation exchange capacity (CEC). CEC impacts how quickly nutrients move through the profile. A soil with a low CEC is much less fertile because it cannot hold on to many nutrients, and they usually contain less clays. If your soil has a low CEC, it is important to apply fertilizer small doses so it does not infiltrate into the groundwater. A soil with a low CEC is less able to hold spilt chemicals.
Soil pH
The soil pH is a measure of soil acidity or alkalinity. pH can range from 1 to 14, with values 0-7 being acidic, and 7-14 being alkaline. Soils usually range from 4 to 10. The pH is one of the most important properties involved in plant growth, as well as understanding how rapidly reactions occur in the soil. .For example, the element iron becomes less available to plants a higher the pH is. This creates iron deficiency problems. Crops usually prefer values between 5.5-8, but the value depends on the crop. The pH of soil comes from the parent material during soil formation, but humans can add things to soils to change them to better suit plant growth. Soil pH also affects organisms. Learn more with this Soil pH activity.
Sorption and Precipitation
Soil particles have the ability to capture different nutrients and ions. Sorption is the process in which one substance takes up or holds another. In this case, soils that have high sorption can hold a lot of extra environmental contaminants, like phosphorus, onto the particles. Soil precipitation occurs during chemical reactions when a nutrient or chemical in the soil solution (water around soil particles) transforms into a solid. This is really important if soils are really salty. Soil chemists study the speed of these reactions under many different conditions.
Soil Organic Matter Interactions
Soil chemists also study soil organic matter (OM), which are materials derived from the decay of plants and animals. They contain many hydrogen and carbon compounds. The arrangement and formation of these compounds influence a soils ability to handle spilt chemicals and other pollutants.

Soil has four major categories of organic matter inside of it, including active and long term types.
Oxidation and Reduction Reactions
Soils that alternate between wet and dry go from having a lot of oxygen to not a lot of oxygen. The presence or absence of oxygen determines how soils chemically react. Oxidation is the loss of electrons, and reduction is the gaining of electrons at the soil surface. These type of reactions occur every day, and are responsible for creating things like rust. Soils, because they contain a lot of iron, can also rust, or if they contain a lot of water, can turn a light gray color. This is partially responsible for all of the different colors that are found, and creates the speckles usually found deeper in the soil.



